
This is a one part lesson called Balancing Chemical Equations, where will learn how to balance chemical equations according to the law of conservation of mass.
So, good luck and have fun learning how to balance chemical equations!
In this video we review the worksheet for lesson 11.

This is a five part lesson called types of chemical reactions.
Each part will consist of a different type of chemical reaction. In each part we will learn about that type of reaction and how to predict the products of a chemical reaction.
So, good luck and have fun learning about types of chemical reactions!

This is a five part lesson called types of chemical reactions.
Each part will consist of a different type of chemical reaction. In each part we will learn about that type of reaction and how to predict the products of a chemical reaction.
So, good luck and have fun learning about types of chemical reactions!
In this video we review the worksheet for Lesson 12.
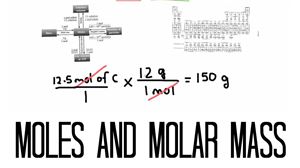
This is a two part lesson called Moles and Molar Mass of Compounds.
The first part is called Moles, and in this part we will learn about the standard unit of measure of chemistry called the Mole.
The second part is called Converting the Mole. In this part we will learn how to convert between the mole and other common units of measure like grams and litres.
So, good luck and have fun learning about Moles and Molar Mass of Compounds!
In this video we review the worksheet for Lesson 13.

This is a three part lesson called Percent Composition, Empirical and Molecular formula.
The first part is called percent composition. In this part we will learn how to determine the percentage of each element in a compound.
The second part is called Empirical Formula. We will learn how to determine the empirical formula of a chemical compound.
The Third part is called molecular formula. We will learn how to determine the molecular formula of a compound based on the empirical formula and the molar mass of the compound.
So good luck and have fun learning about percent composition, empirical and molecular formula!
In this video we review the worksheet for Lesson 14.

This is a three part lesson called stoichiometry, where we will learn how stoichiometry is used to predict amounts of products formed or reactants used up during chemical reactions.
So, good luck and have fun learning about stoichiometry!
In this video we review the worksheet for Lesson 15.
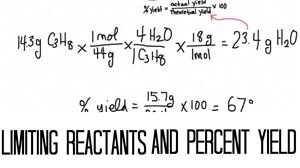
This is a two part lesson called Limiting Reactant and Percent Yield.
The first part is called limiting reactants. In this part we will learn how to determine the limiting reactant in a chemical reaction and then determine the amount of product that could be created.
So good luck and have fun learning about limiting reactants and percent yield!
In this video we review the worksheet for Lesson 16.
